
Stable α-helices typically end with a charged amino acid to neutralize the dipole moment. In addition, the lack of a hydrogen on Proline's nitrogen prevents it from participating in hydrogen bonding.Īnother factor affecting α-helix stability is the total dipole moment of the entire helix due to individual dipoles of the C=O groups involved in hydrogen bonding. Making new lipids at the plasma membrane. Proline also destabilizes α-helices because of its irregular geometry its R-group bonds back to the nitrogen of the amide group, which causes steric hindrance. Question 1 1 out of 1 points A single pass transmembrane protein that is an alpha helix can make a channel. Sorting of proteins to the mammalian endoplasmic reticulum (ER), for secretion or for insertion into the membrane, is mediated by a signal sequence that is characterized by a stretch of 725 mainly apolar residues (Walter and Johnson, 1994). Amino acids whose R-groups are too large (tryptophan, tyrosine) or too small (glycine) destabilize α-helices. The structural integrity of an α-helix is in part dependent on correct steric configuration. The MHC class I HC folds and assembles with 2 m in the lumen of the endoplasmic reticulum (ER). Type I transmembrane proteins are anchored to the lipid membrane with a stop-transfer anchor sequence and have their N-terminal domains targeted to the endoplasmic reticulum (ER) lumen during synthesis (and the extracellular space, if mature forms are located on cell membranes ). Within the lumen, proteins that will be secreted from the cell diffuse into the transitional portion of the ER, a region that is largely free of ribosomes. This regular pattern gives the α-helix very definite features with regards to the thickness of the coil and the length of each complete turn along the helix axis. The O and N atoms of the helix main chain are shown as red and blue balls. The kinemage linked above shows an individual alpha helix, viewed from the N-terminal end to resemble the 'helical wheel' (see figure below). Such a hydrogen bond is formed exactly every 4 amino acid residues, and every complete turn of the helix is only 3.6 amino acid residues. An alpha helix is an element of secondary structure in which the amino acid chain is arranged in a spiral. 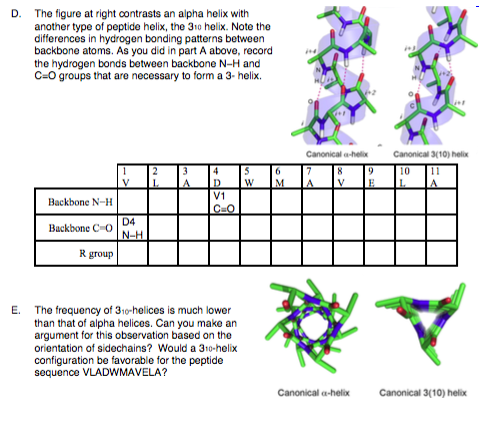
The protein chain runs upward here that is, its N-terminus is at the bottom and its C-terminus at the top. 
Two hydrogen bonds for the same peptide group are highlighted in magenta the H to O distance is about 2 (0.20 nm). This coil is held together by hydrogen bonds between the oxygen of C=O on top coil and the hydrogen of N-H on the bottom coil. Discovery edit Side view of an -helix of alanine residues in atomic detail. \( \newcommand\)Īn α-helix is a right-handed coil of amino-acid residues on a polypeptide chain, typically ranging between 4 and 40 residues.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
